Abstract
Background
Binding of E-selectin (E-sel) to sialyl Lex on the leukemic cell surface activates cell survival pathways and promotes chemotherapy resistance in AML. Expression of the E-sel ligand (E-sel-L) is associated with increased relapse and poor survival. Uproleselan (GMI-1271), a novel E-sel antagonist, disrupts cell survival pathway activation, enhances chemotherapy response and protects from toxicity with improved survival in vivo. We added uproleselan to mitoxantrone, etoposide, cytarabine (MEC) chemotherapy for R/R AML patients (pts) and to cytarabine and idarubicin (7+3) induction for older, treatment naïve (TN) AML pts. Here we report on final outcomes and correlative studies.
Methods
A Phase (Ph) 1/2 trial evaluated safety and efficacy of escalating doses of upro (5-20 mg/kg) combined with MEC in pts with R/R AML. The recommended Ph 2 dose (RP2D) was 10 mg/kg. Ph 2 added pts ≥60 yrs with TN AML treated with upro and 7+3. Uproleselan was given 24 hrs prior, every 12 hrs during and 48 hrs post chemotherapy. Responders could receive consolidation therapy (1 cycle MEC or 1-3 cycles IDAC) with uproleselan. Baseline E-sel-L expression on AML blasts (CD45+,SSC) and leukemic stem cells (LSC, CD34+CD38-CD123+) in blood and bone marrow (BM) was assessed by flow cytometry, for percentage of blasts binding to E-sel-Fc chimeric protein and HECA452 (antibody to sialyl Lex). Post-induction measurable residual disease (MRD-/MRD+) was assessed locally.
Results
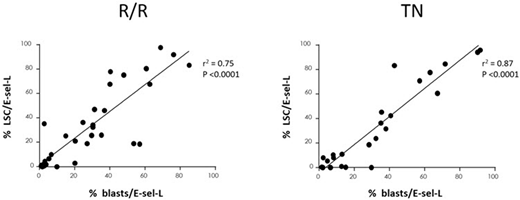
91 pts were enrolled (Ph 1 R/R=19; Ph 2 R/R=47. TN=25). Median age in R/R pts was 59 yrs (26-84) with 22/66 (33%) primary refractory, 22 (33%) CR1<6 months (m); 17% prior SCT; 50% had adverse risk (ELN). Uproleselan was well tolerated, with no increase in adverse events. Grade 3/4 mucositis was 2% at RP2D. R/R CR/CRi rate was 41% (RP2D), 39% (all doses), 30% (primary refractory/CR1<6m). 11/16 (69%) evaluable pts were MRD- . 42/66 (64%) received further anti-leukemic therapy including 17 (26%) SCT. At RP2D, median (95%CI) OS was 8.8 m (5.7-11.4); remission duration was 9.1 m (3.2-15.2); 1-year OS was 35%. For R/R/MRD-, 1-year OS was 73%. E-sel-L was detectable on BM blasts in all 36 evaluable pts: median expression 26% (1-85) of blasts. In a subset of MRD evaluable pts (N=10), E-sel-L expression was higher in those who were MRD- (55% vs 35%). Functional E-sel binding was higher in those achieving CR/CRi (N=14, p=0.003, at 12 hrs; p=0.001 at 48 hrs post uproleselan). In BM blasts (Figure N=36), LSC expression of E-sel-L correlated with blast E-sel-L (R2=0.75, p<0.0001), consistent with hypothesis that E-sel/E-sel-L interaction may be an LSC mechanism of chemoresistance. R/R responders (CR/CRi) also had higher LSC/E-sel-L expression than non-responders (41% [0-98] vs 19% [0.7-83] p=0.06). Median OS for LSC/E-sel-L ≥10% (N=22) vs LSC/E-sel-L <10% (N=14) was 12.7m (8.3-NR) and 5.2 m (2.3-9.4), respectively (p<0.006).
Median age in TN pts was 67 yrs (60-79). 48% had adverse risk (ELN) and 52% secondary AML (sAML). Uproleselan was well tolerated, with no increase in adverse events, no Grade 3/4 mucositis, and mortality 8% (30d) and 12% (60d). CR/CRi was 72% (all), and 69% (sAML). 5/9 (56%) evaluable pts were MRD-. 19/25 (76%) proceeded to further anti-leukemic therapy; 11 (44%) proceeded to SCT. Median (95% CI) EFS, OS, and remission duration were 9.2 m (3.0-12.6), 12.6 m (9.9-NR), and 10.4 m (7.1-17.8) respectively; 1-year OS was 52%. For sAML, median (95% CI) EFS and OS were 7.7 m (1.1-9.5) and 10.5m (4.4-NR), respectively. For TN/MRD-, 1-year OS was 60%. E-sel-L was detectable on BM blasts in all 24 evaluable pts: median expression 31% (2-92) of blasts. In a subset of MRD evaluable pts (N=8), E-sel-L expression was higher in those who were MRD+ (35% vs 8%). In BM blasts (Figure N=24), LSC expression of E-sel-L correlated with blast E-sel-L (R2=0.87, p<0.0001). TN responders (CR/CRi) had LSC/E-sel-L expression similar to non-responders (21% [0-96] vs 21% [0-94] p=NS). Median OS for LSC/E-sel-L ≥10% (N=15) vs LSC/E-sel-L <10% (N=7) was 10.5 m (4.4-NR) and not reached, respectively (p=NS).
Conclusion
The addition of uproleselan to chemotherapy was well tolerated, with low oral mucositis rates, high remission rates, high MRD- and transplant rates, and promising survival outcomes in pts with R/R and TN AML. High E-sel-L expression is associated with improved remission and survival with uproleselan treatment in R/R AML. Phase III studies in pts with R/R and (older) TN AML are underway.
DeAngelo:Shire: Honoraria; Amgen: Consultancy; BMS: Consultancy; ARIAD: Consultancy, Research Funding; Blueprint Medicines: Honoraria, Research Funding; Takeda: Honoraria; Glycomimetics: Research Funding; Novartis Pharmaceuticals Corporation: Consultancy, Honoraria; Amgen: Consultancy; Novartis Pharmaceuticals Corporation: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; Glycomimetics: Research Funding; Blueprint Medicines: Honoraria, Research Funding; Pfizer Inc: Consultancy, Honoraria; Shire: Honoraria; BMS: Consultancy; Takeda: Honoraria; Incyte: Consultancy, Honoraria; Pfizer Inc: Consultancy, Honoraria; ARIAD: Consultancy, Research Funding. Jonas:Glycomimetics: Research Funding; Genentech/Roche: Research Funding; Celgene: Consultancy, Research Funding; Accelerated Medical Diagnostics: Research Funding; Daiichi Sankyo: Research Funding; Incyte: Research Funding; Esanex: Research Funding; Tolero: Consultancy; Kalobios: Research Funding; LP Therapeutics: Research Funding; Amgen: Consultancy; Pharmacyclics: Research Funding; Forma: Research Funding; AbbVie: Consultancy, Research Funding. Liesveld:Onconova: Other: DSMB; Abbvie: Honoraria. Bixby:GlycoMimetics: Research Funding. Advani:Glycomimetics: Consultancy; Novartis: Consultancy; Amgen: Research Funding; Pfizer: Honoraria, Research Funding. Marlton:GlycoMimetics: Research Funding; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees; Astellas: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees. O'Dwyer:Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Research Funding; Onkimmune: Equity Ownership, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Research Funding; Glycomimetics: Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees. Fogler:GlycoMimetics: Employment, Equity Ownership. Wolfgang:GlycoMimetics: Employment, Equity Ownership. Magnani:GlycoMimetics: Employment, Equity Ownership, Membership on an entity's Board of Directors or advisory committees. Thackray:GlycoMimetics: Employment, Equity Ownership. Becker:GlycoMimetics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal